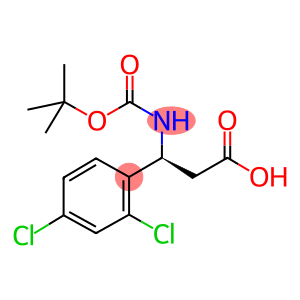
(S)-3-TERT-BUTOXYCARBONYLAMINO-3-(2,4-DICHLORO-PHENYL)-PROPIONIC ACID
Boc-S-3-Amino-3-(2,4-dichloro-phenyl)-propionic acid
CAS: 499995-81-2
Molecular Formula: C14H17Cl2NO4
(S)-3-TERT-BUTOXYCARBONYLAMINO-3-(2,4-DICHLORO-PHENYL)-PROPIONIC ACID - Names and Identifiers
(S)-3-TERT-BUTOXYCARBONYLAMINO-3-(2,4-DICHLORO-PHENYL)-PROPIONIC ACID - Physico-chemical Properties
| Molecular Formula | C14H17Cl2NO4 |
| Molar Mass | 334.2 |
| Density | 1.321±0.06 g/cm3(Predicted) |
| Boling Point | 475.2±45.0 °C(Predicted) |
| pKa | 4.22±0.10(Predicted) |
| Storage Condition | 2-8°C |
(S)-3-TERT-BUTOXYCARBONYLAMINO-3-(2,4-DICHLORO-PHENYL)-PROPIONIC ACID - Introduction
Boc-S-3-Amino-3-(2,4-dichloro-phenyl)-propionic acid is an organic compound. Its chemical formula is C16H18Cl2N2O4S and its molecular weight is 403.29g/mol.
Nature:
Boc-S-3-Amino-3-(2,4-dichloro-phenyl)-propionic acid is a colorless to pale yellow solid, soluble in some organic solvents such as dimethylthionamide. Its melting point is about 135-138°C.
Use:
Boc-S-3-Amino-3-(2,4-dichloro-phenyl)-propionic acid can be used as an organic synthetic intermediate, which is commonly used in pharmaceutical research and biochemical experiments. As an amino acid derivative, it may be used in the synthesis of biologically active molecules, such as drug candidate compounds.
Method:
The preparation of Boc-S-3-Amino-3-(2,4-dichloro-phenyl)-propionic acid is usually carried out by chemical synthesis methods. Specific synthetic steps involve a series of organic reaction steps, including substitution and addition of protecting groups, etc.
Safety Information:
Safety data on Boc-S-3-Amino-3-(2,4-dichloro-phenyl)-propionic acid are limited. As a chemical substance, use should follow laboratory safety procedures, wear appropriate personal protective equipment, and avoid contact with skin, eyes and respiratory tract. In addition, due to its organic synthesis characteristics, it may produce harmful gases, vapors and fumes, and attention should be paid to well-ventilated conditions during operation. In case of any discomfort or accident, the doctor should be referred to promptly and relevant materials should be provided.
Nature:
Boc-S-3-Amino-3-(2,4-dichloro-phenyl)-propionic acid is a colorless to pale yellow solid, soluble in some organic solvents such as dimethylthionamide. Its melting point is about 135-138°C.
Use:
Boc-S-3-Amino-3-(2,4-dichloro-phenyl)-propionic acid can be used as an organic synthetic intermediate, which is commonly used in pharmaceutical research and biochemical experiments. As an amino acid derivative, it may be used in the synthesis of biologically active molecules, such as drug candidate compounds.
Method:
The preparation of Boc-S-3-Amino-3-(2,4-dichloro-phenyl)-propionic acid is usually carried out by chemical synthesis methods. Specific synthetic steps involve a series of organic reaction steps, including substitution and addition of protecting groups, etc.
Safety Information:
Safety data on Boc-S-3-Amino-3-(2,4-dichloro-phenyl)-propionic acid are limited. As a chemical substance, use should follow laboratory safety procedures, wear appropriate personal protective equipment, and avoid contact with skin, eyes and respiratory tract. In addition, due to its organic synthesis characteristics, it may produce harmful gases, vapors and fumes, and attention should be paid to well-ventilated conditions during operation. In case of any discomfort or accident, the doctor should be referred to promptly and relevant materials should be provided.
Last Update:2024-04-09 21:54:55
Supplier List
Product Name: Boc-S-3-Amino-3-(2,4-dichloro-phenyl)-propionic acid Request for quotation
CAS: 499995-81-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 499995-81-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: (S)-3-((tert-Butoxycarbonyl)amino)-3-(2,4-dichlorophenyl)propanoic acid Visit Supplier Webpage Request for quotationCAS: 499995-81-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: BOC-(S)-3-AMINO-3-(2,4-DICHLORO-PHENYL)-PROPIONIC ACID Request for quotation
CAS: 499995-81-2
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 499995-81-2
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: BOC-(S)-3-AMINO-3-(2,4-DICHLORO-PHENYL)-PROPIONIC ACID Visit Supplier Webpage Request for quotation
CAS: 499995-81-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 499995-81-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: Boc-S-3-Amino-3-(2,4-dichloro-phenyl)-propionic acid Request for quotation
CAS: 499995-81-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 499995-81-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: (S)-3-((tert-Butoxycarbonyl)amino)-3-(2,4-dichlorophenyl)propanoic acid Visit Supplier Webpage Request for quotationCAS: 499995-81-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: BOC-(S)-3-AMINO-3-(2,4-DICHLORO-PHENYL)-PROPIONIC ACID Request for quotation
CAS: 499995-81-2
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 499995-81-2
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: BOC-(S)-3-AMINO-3-(2,4-DICHLORO-PHENYL)-PROPIONIC ACID Visit Supplier Webpage Request for quotation
CAS: 499995-81-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 499995-81-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



